More About The Serval
Serval Cat
(Felis serval)
 Servals have relatively the longest legs of all cats, an adaptation that is not for fast running, as is often asserted, but which seems to be primarily related to the problem of hunting small or medium prey in tall grass. Scent-oriented predators can follow the trails of small animals but the serval, like other cats, depends on sight and hearing to detect prey. The former sense is severely impeded in tall grass so the serval is left with hearing, which is very acute and finds expression in the enormous ears. To get an accurate “fix” by means of sound alone the detector needs to gain elevation and this is likely to be the prime reason for the Servals tallness. The long limbs assist spectacular vertical jumps and lightning jabs with the forepaws, but increased striking range is itself dependent on the elevation of the predator’s vantage point. The Servals’ ears are their most prominent and highly characteristic features. Their backs, like those of the lion and leopard, are decorated with white blobs surrounded by black margins. The ears’ role in signaling, however, is evidently subsidiary to their primary function as sound funnels and they are not flickered about in the way a caracal’s ears are. In the more westerly part of the equatorial zone the serval cat’s pattern is dimorphic. A fine freckled form (servaline) and a bold black-spotted form occur in about equal ratios in south-western Uganda and in eastern Zaire, a region in which it is common to find both
Servals have relatively the longest legs of all cats, an adaptation that is not for fast running, as is often asserted, but which seems to be primarily related to the problem of hunting small or medium prey in tall grass. Scent-oriented predators can follow the trails of small animals but the serval, like other cats, depends on sight and hearing to detect prey. The former sense is severely impeded in tall grass so the serval is left with hearing, which is very acute and finds expression in the enormous ears. To get an accurate “fix” by means of sound alone the detector needs to gain elevation and this is likely to be the prime reason for the Servals tallness. The long limbs assist spectacular vertical jumps and lightning jabs with the forepaws, but increased striking range is itself dependent on the elevation of the predator’s vantage point. The Servals’ ears are their most prominent and highly characteristic features. Their backs, like those of the lion and leopard, are decorated with white blobs surrounded by black margins. The ears’ role in signaling, however, is evidently subsidiary to their primary function as sound funnels and they are not flickered about in the way a caracal’s ears are. In the more westerly part of the equatorial zone the serval cat’s pattern is dimorphic. A fine freckled form (servaline) and a bold black-spotted form occur in about equal ratios in south-western Uganda and in eastern Zaire, a region in which it is common to find both
morphs in a single litter. In drier areas further south and east large-spotted morphs are the dominant or exclusive type.
Melanistic Servals are common in the highland areas of Kenya and Ethiopia and have also been recorded from northern Tanzania, Tsavo, South Ankole, and Karamoja. Like those of leopards and cheetahs, the Serval’s spots are probably the product of a process of amalgamation, which suggests that the “servaline” freckle pattern represents a phylogenetically older type of organization. At one time freckled skins were mistakenly assigned to distinct species: F. brachyura and F. servalina. The predominance of the bold spotted morph in south-eastern Africa suggests that there has been stronger selection for this type in the drier grasslands than in the more moist equatorial regions. What this selective pressure might be is difficult to imagine but there is some correlation between humidity and tonal effect  (Gloger’s rule) in that melanistic servals and servalines, which are darker in overall tone, come from moist areas. Studying Servals in the Aberdare National Park, York (1973), describes animals with varying degrees of melanism as well as all-black animals.
(Gloger’s rule) in that melanistic servals and servalines, which are darker in overall tone, come from moist areas. Studying Servals in the Aberdare National Park, York (1973), describes animals with varying degrees of melanism as well as all-black animals.
Servals are found in all the grass savannas of Africa and are common in sub-alpine habitats including bamboo and
forest evnironments as long as they are well interspersed with grassy glades or moorland. At lower altitudes, they are abundant along the margins of forest galleries and of reed beds and marshes. They have adapted well to the cultivation fallow mosaic that is now widespread over the moister parts of Africa. A very wide range of small mammals, birds, reptiles and insects (notably termites and grasshoppers) have been recorded in their diet but they may, at times, partly abandon predaceous habits. Verschuren (1958) found that four out of seven stomachs of Garamba Servals contained mainly vegetable matter and Rahm and Christiaensen (1963) noted bananas and avocado. My own captives sometimes ate quantities of
green grass but this is common in many felid and canid species. Judging by published records, hares and rodents are the most common foods with murid species such as Mastomys, Arvicanthis, Lemniscomys and Dasymys, mole rats, Tachyoryctes and Cryptomys, ground squirrels, Xerus erythropus and cane rats, Thryonomys, all recorded.
Both diurnal and nocturnal prey species are represented and this accords with the observed activity of Servals, which is mainly crepuscular and nocturnal but extends into the day in some localities during the wet season. Where human settlement is intensive, it is generally wholly nocturnal. Most prey is killed on the ground but serval has been reported pursuing a hyrax up a tree. Nonetheless, it is not normally arboreal. Verheyen (1951) reported Servals killing duikers and the young of larger species of antelopes, such as oribi and bushbuck but Rahm noted that adult duikers are only successfully hunted when there are two Servals, and general observations of single cats in fruitless pursuit of small
antelopes gives some credence to this. The largest recorded prey is a female impala on which two Servals were seen
one early morning; as the animal was killed in long grass she had presumably been ambushed. When prey is fairly large or likely to bite back, the serval springs high and strikes hard with all four feet and delivers a deep and accurate  bite. It is interesting that this technique, which appears to be adapted to small prey, is not greatly modified for larger victims and York (1973) describes a Serval circling for a few seconds and repeating its high spring and bite when it was dislodged by the violence of a baby gazelle’s struggles. This 7kg fawn was only killed after several minutes of effort.
bite. It is interesting that this technique, which appears to be adapted to small prey, is not greatly modified for larger victims and York (1973) describes a Serval circling for a few seconds and repeating its high spring and bite when it was dislodged by the violence of a baby gazelle’s struggles. This 7kg fawn was only killed after several minutes of effort.
Prey that is first identified by sight is stalked by eye to as close as possible, the cat crouching low on the ground, perhaps raising its head cautiously if the prey disappears from sight. A short rush may follow but chases are seldom sustained for more than a few tens of meters. Birds are sprung upon with a prodigious leap or in a short charge, grabbed with the claws, and instantly immobilized with a bite in the neck. Rahm and Christiaensen saw Servals excavating Tachyoryctes by scratching away their earth mounds and digging into the short burrow. Then they have been seen to leap up in the air and alight on the mole rat. The blesmol (Cryptomys) on the other hand has a very long burrow but hastens to repair the break if an entrance is unplugged. In this case Verheyen reported the serval scratching mounds away and awaiting the arrival of the blesmol which it then seized in its teeth. I have often watched Servals hunting very small prey that was totally hidden in grass, in which case the cats listened very intently and then  hooked down at the rustling.
hooked down at the rustling.
If contact is not made with the first jab, the Serval continues with a very fast succession of bouncing strikes with the neck erect, chin in chest, and tail vertical. This is a searching technique for which the long loose digits and its strongly curved claws are particularly well adapted. If the prey is hooked it may be tossed up and then pounced upon or the cat
may hold the rat down while it bites the back of its neck and then lets go, watching it intently. When my hand-reared female discovered small Typhlops snakes she would kill them with repeated strokes of her paw but very seldom mouthed or ate them, although this was seen on one occasion. Rustling in leaf litter or in short or flattened grass always precipitates an attack and one must be careful with pet Servals not to let one’s fingers fidget beneath cover at ground level or the results will be very painful. Even when the Serval is hungry it may choose to play with the dead or dying animal, seizing it in the mouth, and tossing it into the air and often standing on the hind legs to swipe at it as it falls. This is particularly noticeable whenever the prey is a bird. Servals are accomplished pluckers which suggests that birds are a regular item of their diet. Quails, Coturnix, spur fowl Pternistes, guineafowis, Numidia, and bustards, Otidae have been recorded in their diet. My own captives ate a variety of species and especially relished doves but always refused duck.
The plucking technique is also applied but with less thoroughness to hares and it is interesting to see that when birds or hares are lifted in the mouth and tossed, the tossing movements often change imperceptibly into deliberate plucking. In fact the two activities may be homologous, for plucking is unlike any form of chewing, biting, or tearing and requires a different type of motor pattern. As playing with maimed or dead prey is universal among felids whereas skilful plucking is not, it is possible that Servals and caracals developed their plucking expertise by the elaboration of an innate action pattern. Even captives reared in isolation display the capacity to pluck so it is not a learned skill. De La Fuente (1970) mentions Servals eating fish and my own captives relished them and would hook live fish out of a basin one after the other with fast jabs of one foreleg and a deft scoop of the wrist which sent the fish flying. When the prey animal is too large to be eaten at one sitting the Serval may cache it. I watched my captive female hide the back
part of a hare she had half eaten by scratching leaves, dead grass and other detritus over it with both front and back paws. The same female would sometimes make kicking or raking movements in the litter when she was given a particularly large or heavy piece of meat or, at times, when she had been approached suddenly and silently and therefore appeared to be surprised or nervous. When this animal was fed at a time when a strange male Serval was newly housed nearby, she carried her meat continuously for fifteen minutes, pacing up and down her run with her eyes staring and making jerky movements. Finally the meat was eaten without play in the most secluded corner of her run.
The smaller felids seldom share their spoils so it is all the more intriguing to consider the status of those Servals seen sharing or participating in a single large kill. From observation of captives it seems very unlikely that adult females would ever associate and females only tolerate a male during estrus, an association that does not last for very long and tends to be preoccupied with sexual activity. Juveniles or a mother and her young are not as well equipped to tackle larger prey as adult males, which are appreciably heavier, stronger, and more powerfully built. The extension of tall grass growth that is brought on by the rains must alter the Servals pattern of land tenure and the extension of suitable 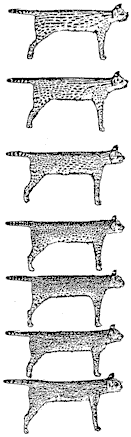 cover presumably increases the potential range of individuals considerably. Rodent populations generally increase during the rains but are more dispersed. The extension of long grass areas, however, would undoubtedly render large prey more vulnerable to the Serval and there is a greater likelihood of antelopes blundering into an ambush. Such circumstances could offer an incentive to Servals.
cover presumably increases the potential range of individuals considerably. Rodent populations generally increase during the rains but are more dispersed. The extension of long grass areas, however, would undoubtedly render large prey more vulnerable to the Serval and there is a greater likelihood of antelopes blundering into an ambush. Such circumstances could offer an incentive to Servals.
There also could be advantages for Servals to form associations which could be better able to handle larger prey. It is thought that there may be a latent sexual cycle in male domestic cats which corresponds to that of the female (Aronson and Cooper, 1966). In the case of the Serval cat there is some evidence from Uganda of biannual phasing of the female cycle which is timed to coincide with the two driest times of the year. This suggests that the Uganda Servals mate while their ideal habitat is most restricted and their range most fragmented. It would be very interesting to learn whether males are any more or less tolerant of one another during the wet season when they may be relatively free of a sexual, and perhaps territorial, role, and whether neighboring males ever form hunting partnerships during the rains. In Kenya, York (1973) found evidence for territories in the form of urine squirts, dung deposits, and scratching on termitaries, rocks, and trees. He described individuals traveling repeatedly over established tracks and routinely turning at the same point.
Sometimes confrontations were suggested by the footprints of two Servals pacing up and down at what appeared to be a territorial boundary and he watched one incident in which a Serval approached a clearing and started calling, whereupon another Serval, a male, was seen to enter the clearing from a different direction, urinate, then scratch at the foot of a low bush. When the animal that had been calling appeared, this male arched its back with its tail up and made threatening calls. The other cat responded to the threats for half a minute but with lowered fore-quarters and when the male approached it moved off with frequent backward glances and growls. The male sniffed the spot where the departed had stood and after scratching the ground defecated and followed the retreating cat. It is interesting that captive Serval cats exhibit a sexual difference in their defecating habits, females using the same place while males drop their dung in an apparently more random way. The habitual spraying of particular posts by male Servals leaves a greasy and powerfully scented deposit and when repeated several times causes green growth to wilt, thus increasing the conspicuousness of the mark.
The postures of Servals are very exaggerated. In aggressive encounters the ears form a flat plate with a characteristic curl to their decorated tips. With heightened intensity, the back is arched and the Serval stands on greatly elongated legs in a laterally presented stance. The dorsal fur bristles, the eyes blaze, and ferocious explosive barks and growls are interspersed with long-reaching slashes of the forepaws. If one cat approaches another with the body half crouched 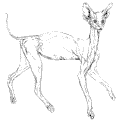 and tense this indicates a readiness to flee. The head may be raised and lowered on the long neck several times before there is a further advance and the eyes remain wide open, the ears pricked and alert. A curious swallowed mew seems to signify friendly recognition and may be followed by an abrupt about turn and a shiver-display of the vertical tail, which is the most conspicuous signal in the Servals repertory. A female betrays the onset of her estrus by a very short, sharp meow which has considerable carrying power and is repeated in bouts. When approached by a male she may purr and raise her chin as she rubs the side of her mouth and cheeks against him or on intervening wire.
and tense this indicates a readiness to flee. The head may be raised and lowered on the long neck several times before there is a further advance and the eyes remain wide open, the ears pricked and alert. A curious swallowed mew seems to signify friendly recognition and may be followed by an abrupt about turn and a shiver-display of the vertical tail, which is the most conspicuous signal in the Servals repertory. A female betrays the onset of her estrus by a very short, sharp meow which has considerable carrying power and is repeated in bouts. When approached by a male she may purr and raise her chin as she rubs the side of her mouth and cheeks against him or on intervening wire.
When my solitary female was in estrus, she would rub the sides of her cheeks against my knee, hand, or face and salivate copiously out of the corners of her mouth, so that one became quite wet with spit. She also lathered herself with saliva but actually used her long tongue while self-grooming. She would stand with her snowy-white chin raised and then bring it down with a rolling, rubbing movement that smeared saliva up her cheek and on to her head and ears. This downward rub was sometimes correlated with dropping on to her chest but with the hindquarters still raised, approximating to the submissive posture with which a female solicits a male.
More commonly the female stands and, after rubbing the cheek and neck, walks on leaning heavily against the male, a human substitute, an intervening wire, or an inanimate object. As soon as the rump is reached, the vertical tail shivers and vibrates, whereupon she lies down or turns round very sharply to repeat the performance, purring loudly all the while. When a captive female is in estrus and on her own, she intersperses bouts of yowling with interminable pacing and frequent but sparing urine squirting at particular points. The vertical tail is always vibrated when this is done and she may smell or even rub her head on the same spot or may lie and roll displaying the almost incandescent white of her spotted belly in the typical felid submission posture. She may rake her claws down a particular tree trunk. My female used different posts for squirting and raking, but both activities are much more common in males. The observation of a dominant male raking over a spot that had been scented by an intruder betrays the aggressive symbolism of raking and I have occasionally noticed captives turn round after seizing proffered food and rake the ground with both fore and back legs, a reaction that was generally associated with uncertainty or traces of hostility at my own approach. Because solitary animals avoid sharing their food, unfinished meals might elicit displaced aggression so that even the act of raking vegetation over left-leftovers might have originated in aggressive no-sharing impulses.
Like other cats, Servals may lie up on an eminence such as a termitary, usually during the morning, where they can sunbathe for a while before retiring to a concealed retreat. I have found them lying up spots under flattened reeds or  tall grass and suspect that Servals may keep circulating, for forms do not show signs of lengthy occupation. The young are reported to be born in a well-hidden retreat in dense vegetation, in a hollow tree, or down a hole and Verheyen (1951) suggested that they leave the shelter earlier than most cats. Gestation lasts 64-78 days and eight records of births from Uganda and eastern Zaire suggest two birth periods during the wet season, one in March-April and the other between September and November. This accords well with data from the estrous periods exhibited by captive females, which reach their peak in the dry periods of December-January and between June and August. Two or three young (range one to five) are born, blind and helpless. Their tail is short and ears relatively short and folded (see drawing) but the growth of the ears is astonishingly rapid. The young attempt to accompany the mother at about three weeks but a captive mother has been seen returning her young to the nest whenever they tried to wander (Boston, personal communication). Up to about three months old, they make a curious chirping noise. If left on their own they hide and freeze at any disturbance and remain absolutely still although following every movement with the eyes.
tall grass and suspect that Servals may keep circulating, for forms do not show signs of lengthy occupation. The young are reported to be born in a well-hidden retreat in dense vegetation, in a hollow tree, or down a hole and Verheyen (1951) suggested that they leave the shelter earlier than most cats. Gestation lasts 64-78 days and eight records of births from Uganda and eastern Zaire suggest two birth periods during the wet season, one in March-April and the other between September and November. This accords well with data from the estrous periods exhibited by captive females, which reach their peak in the dry periods of December-January and between June and August. Two or three young (range one to five) are born, blind and helpless. Their tail is short and ears relatively short and folded (see drawing) but the growth of the ears is astonishingly rapid. The young attempt to accompany the mother at about three weeks but a captive mother has been seen returning her young to the nest whenever they tried to wander (Boston, personal communication). Up to about three months old, they make a curious chirping noise. If left on their own they hide and freeze at any disturbance and remain absolutely still although following every movement with the eyes.
If reared from infancy they can become handsome pets and are a favorite zoo animal. They will breed in captivity and can live to at least thirteen years. They are probably subject to most feline diseases and a male I kept died after several days of intense irritation of the skin, during which he literally plucked out all his hair. It may have been co-incidental but the condition began two days after having received a bite from an adult Cricetomyt gambianut that he killed and ate. Leopards have been seen to pursue Servals and not infrequently dogs bring them to bay by driving them up a tree, although it is apparently not unknown for a serval to kill a dog. The skin of this species is sometimes sold as a “young leopard” or cheetah and commands a good price in the local skin trade; as a result this cat is no longer found in heavily populated areas.
